Annual Summary of Disease Activity:
Disease Control Newsletter (DCN)
Related Topics
Contact Info
Campylobacteriosis, 2018
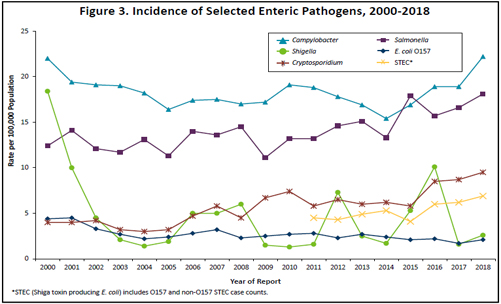
There were 1,238 culture-confirmed Campylobacter cases reported in 2018 (22.2 per 100,000 population). This is an 18% increase over the 1,049 cases reported in 2017, and a 32% increase from the annual median of 939.5 cases reported from 2008 to 2017 (range, 834 to 1,049) (Figure 3). In 2018, 48% of cases occurred in people who resided in the metropolitan area. Of the 1,178 Campylobacter isolates confirmed and identified to species by MDH, 83% were C. jejuni and 12% were C. coli.
The median age of cases was 36 years (range, 3 months to 95 years). Forty-two percent were between 20 and 49 years of age, and 9% were ≤5 years of age. Fifty-five percent were male. Fifteen percent were hospitalized; the median length of hospitalization was 4 days. Forty-five percent of infections occurred during June through September. Of the 1,126 cases for whom data were available, 233 (21%) reported travel outside the United States during the week prior to illness onset. The most common travel destinations were Europe (n=67), Mexico (n=52), Asia (n=47), Central or South America or the Caribbean (n=36), Africa (n=17), and the Middle East (n=12).
Seven outbreaks of Campylobacter infections were identified. Three outbreaks were due to foodborne transmission. One outbreak was associated with chicken liver pâté served at a restaurant, one outbreak was associated with a restaurant with an unknown vehicle of transmission, and one multistate outbreak was associated with consumption of chicken livers. An additional probable foodborne outbreak was likely caused by chicken wings served at a restaurant. Two animal contact outbreaks were identified; the vehicle of transmission was contact with puppies for both outbreaks. One outbreak of Campylobacter infections was associated with a child care facility, but the route of transmission was not confirmed.
A primary feature of public health importance among Campylobacter cases was the continued presence of Campylobacter isolates resistant to fluoroquinolone antibiotics (e.g., ciprofloxacin), which are commonly used to treat campylobacteriosis. In 2018, the overall proportion of quinolone resistance among Campylobacter isolates tested (n=129) was 40%. However, 89% of Campylobacter isolates from patients with a history of foreign travel during the week prior to illness onset, regardless of destination, were resistant to fluoroquinolones. Twentyone percent of Campylobacter isolates from patients who acquired the infection domestically were resistant to fluoroquinolones.
In 2009, a culture-independent test (CIDT) became commercially available for the qualitative detection of Campylobacter antigens in stool. In 2018, 74 patients were positive for Campylobacter by an antigen detection CIDT conducted in a clinical laboratory. However, only 17 (23%) of the specimens were subsequently culture-confirmed. Beginning In 2015, some clinical laboratories in Minnesota began testing stool specimens with PCR-based gastrointestinal pathogen panels, another type of CIDT. In 2018, 1,235 patients were positive for Campylobacter by a PCR gastrointestinal panel; 955 (77%) of these specimens were culture-confirmed. Only culture-confirmed cases met the surveillance case definition for inclusion in MDH case count totals.
- For up to date information see>> Campylobacteriosis (Campylobacter)
- Full issue>> Annual Summary of Communicable Diseases Reported to the Minnesota Department of Health, 2018