Respiratory Syncytial Virus (RSV)
Infectious Respiratory Illness
- Infectious Respiratory Illness Home
- Viral Respiratory Illness in Minnesota (Data & Statistics)
- For Health Professionals
Related Topics
Contact Info
Respiratory Syncytial Virus (RSV) for Health Professionals
On this page:
Reporting
Infection prevention and control
Immunization options
Resources for healthcare providers
Reporting
- Reporting Respiratory Syncytial Virus (RSV)
As of Oct. 1, 2023, laboratory-confirmed RSV in all hospitalized patients who are Minnesota residents should be reported to the Minnesota Department of Health (MDH). - LTC Facility Influenza and RSV Report Form 2025-2026
MDH collects reports of outbreaks of RSV and influenza among residents living in long term care facilities. Please follow this link for more information and to fill out our online reporting form.
Infection prevention and control
Information for health professionals, schools, and childcare providers on RSV epidemiology, infection prevention, and control measures.
- American Academy of Pediatrics Red Book: RSV
American Academy of Pediatrics (AAP) reference with information on clinical manifestations, epidemiology, infection prevention, and control measures. - Hennepin County infectious diseases in childcare settings and schools manual
Section 6 contains fact sheets for school and childcare providers on diseases including RSV. Information on signs and symptoms, spread, exclusion, and prevention and control.
Immunization options
MDH endorses the immunization schedules from the American Academy of Pediatrics (AAP), the American Academy of Family Physicians (AAFP), and the American College of Obstetricians and Gynecologists (ACOG). For more information refer to Immunization Information for Healthcare Providers.
Minnesota Respiratory Syncytial Virus (RSV) Vaccination Guide (PDF)
Information for the 2025-26 respiratory season for providers who handle or administer RSV vaccine. This guide summarizes vaccination recommendations, gives tips on vaccine administration, storage, and handling, and provides additional resources.
Due to continued RSV circulation at the end of the 2025-26 season, MDH has recommended extending the use of RSV monoclonal antibody immunization for infants through April 30. For more information refer to Health Advisory: Health Advisory: MDH Extends Date for Use of RSV Monoclonal Antibodies Through April 30, 2026 (PDF).
CDC recommends RSV vaccination for all adults 75 years of age and older and for adults 50-74 who are at increased risk of severe RSV disease. Adults who could benefit from vaccination due to higher risk of severe disease caused by RSV are those with underlying medical conditions, frailty or advanced age, and residents in long-term care facilities. Eligible adults can get an RSV vaccine at any time, but the best time to get vaccinated is in late summer and early fall before RSV usually starts to spread in communities.
Vaccine resources for older adults
- Immunize.org: RSV (Respiratory Syncytial Virus)
Includes healthcare professional organizations’ vaccine recommendations, Vaccine Information Sheets (VISs), and FDA package inserts. - VaccineInformation.org: RSV (Respiratory Syncytial Virus)
Provides RSV overview and partner resources. - Immunize.org: RSV Vaccine Information Statements (VIS)
- Vaccine Integrity Project: 2025-2026 Respiratory Season
- CDC: Clinical Overview of RSV
To prevent severe RSV disease in infants, either maternal RSV vaccination or infant immunization with RSV monoclonal antibody is recommended. Most infants will not need both.
Vaccination during pregnancy
- Pfizer Abrysvo is the only RSV vaccine recommended during pregnancy.
- One dose should be given during weeks 32 through 36 of pregnancy, administered September through January (to protect infants born October through March).
- No additional doses should be given in subsequent pregnancies; the infant should receive a monoclonal antibody instead.
Monoclonal antibody prophylaxis
- Nirsevimab (also known as Beyfortus) and clesrovimab (also known as Enflonsia) are long-acting RSV monoclonal antibodies recommended for infants/toddlers to prevent lower respiratory tract disease from RSV.
- Monoclonal antibodies should be given:
- In the first week of life for infants born shortly before and during the season.
- Shortly before the start of the RSV season for infants aged less than 8 months (October through March*).
- For nirsevimab only: Shortly before the start of the RSV season for children aged 8 to 19 months who are at increased risk of severe RSV disease or are American Indian and Alaska Native (October through March*).
*For the 2025-26 season refer to Health Advisory: Health Advisory: MDH Extends Date for Use of RSV Monoclonal Antibodies Through April 30, 2026 (PDF) for guidance on changes in recommendations.
- The dosage is dependent on the product used:
- Nirsevimab is weight dependent:
- Infants less than 5kg (11 pounds) get 50mg.
- Infants 5kg (11 pounds) and over get 100mg.
- Clesrovimab is 105mg for all infants regardless of weight.
- Nirsevimab is weight dependent:
- The supply of monoclonal antibody products is projected to be adequate for the 2025-26 season.
Vaccine resources for infant and pregnant people
- Immunize.org: RSV (Respiratory Syncytial Virus)
Includes healthcare professional organizations’ vaccine recommendations, Vaccine Information Sheets (VISs), and FDA package inserts. - VaccineInformation.org: RSV (Respiratory Syncytial Virus)
Provides RSV overview and partner resources. - AAP: Nirsevemab Administration Visual Guide (PDF)
- Vaccine Integrity Project: 2025-2026 Respiratory Season
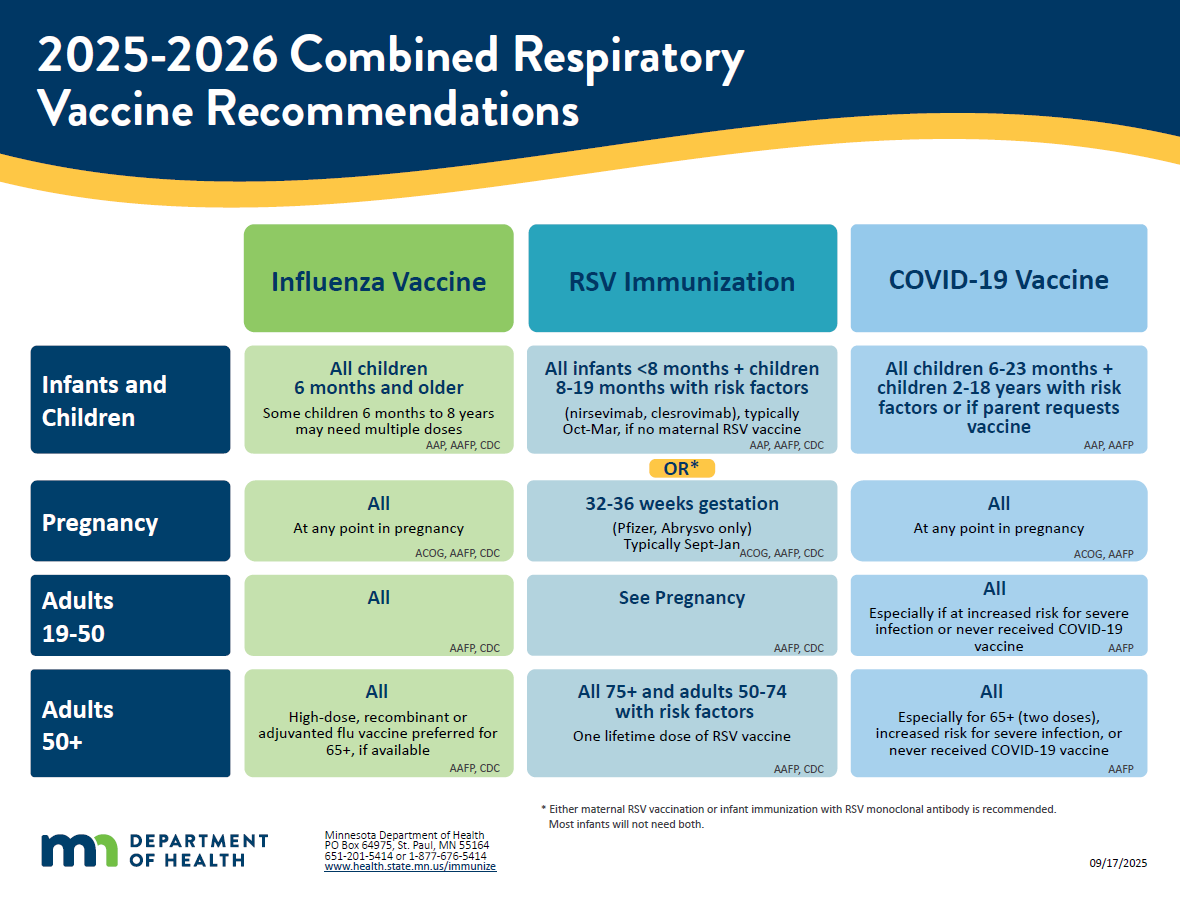
2025-2026 Combined Respiratory Vaccine Recommendations (PDF)
Handout version of the respiratory vaccine recommendations above.
Resources for healthcare providers
These resources can be used to explain and encourage vaccination and immunization for pregnant people and/or infants as well as help them keep record of it. Communication between the pregnant person and infant providers will be important to ensure that all infants are protected.
 | Abrysvo: Respiratory Syncytial Virus (RSV) Vaccine Card (one card) (PDF)
|
 | RSV Immunization Card (one card) (PDF)
|
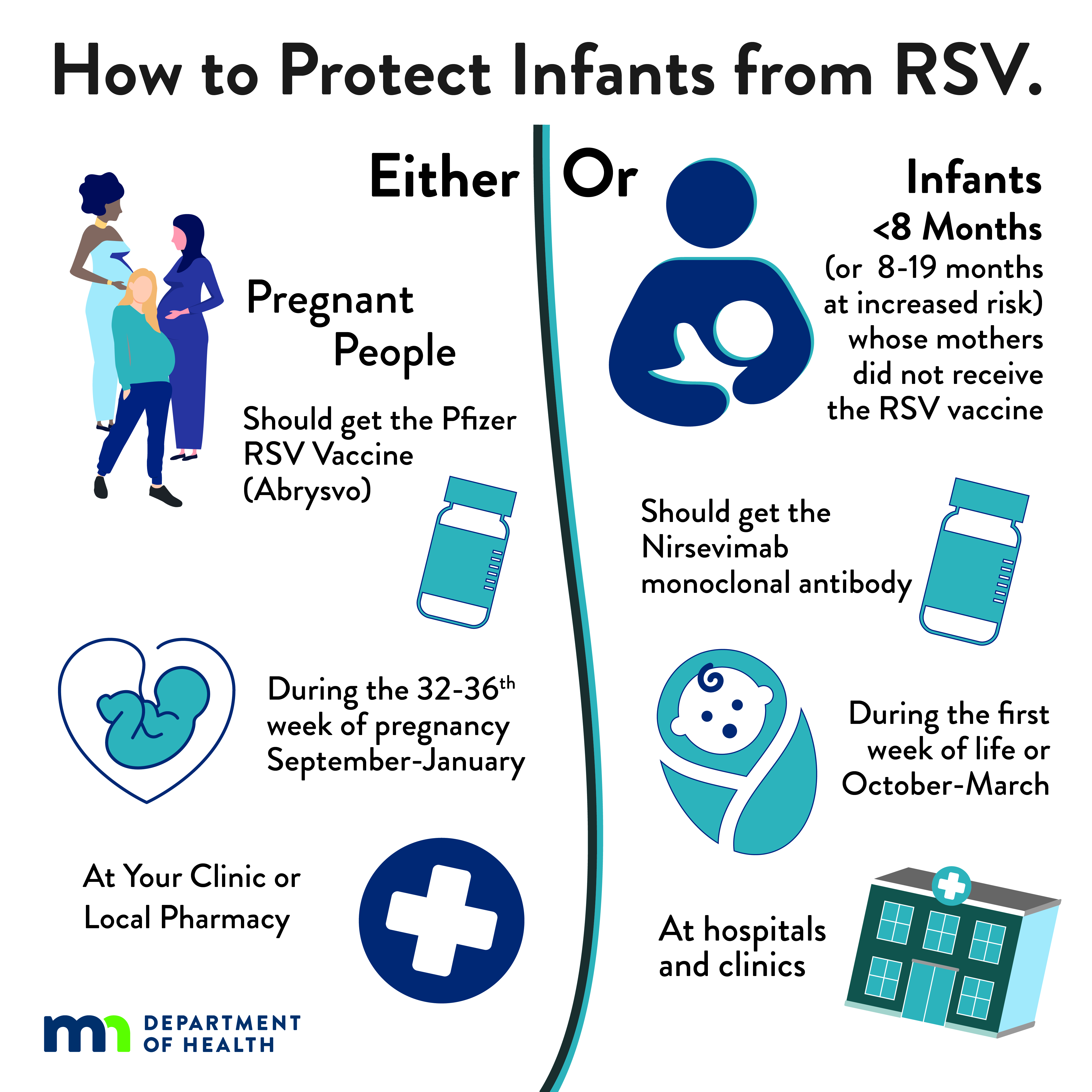 | Infographic: How to Protect Infants from RSV: Infants or Pregnant People (JPG) Social media infographic explaining both the RSV vaccine for pregnant people and the immunization for infants. |
