STD Information For Health Professionals
- STD: Health Professionals Home
- FAQs about STD & HIV Reporting
- Confidential STD Case Report Form
- Expedited Partner Therapy (EPT)
- Demystifying HIV and STIs Webinar Series
Related Topics
Contact Info
Expedited Partner Therapy (EPT) for infections due to Chlamydia trachomatis, Neisseria gonorrhoeae, and/or Trichomonas vaginalis
Guidance for Health Professionals in Minnesota
The intended audience for the guidance herein is healthcare professionals in the state of Minnesota. This document provides up to date information on the most appropriate indications, medications, implementation strategies, and counseling procedures recommended by CDC and MDH to maximize patient and public health benefit of EPT while minimizing risk. For additional questions about EPT or this guidance, please use the IDEPC Comment Form or call 651-201-5414. Legal advice is not provided within this guidance. Consultation with your and/or your organization’s legal counsel is recommended if there are questions about the law, rules, statutes, and practices presented herein.
Introduction to Expedited Partner Therapy (EPT)
Expedited partner therapy (also known as EPT, expedited partner treatment, or partner-delivered partner treatment) is a harm reduction strategy and is defined as the practice of treating the sexual partners of patients diagnosed with certain qualifying sexually transmitted infections by providing antimicrobial treatment for the partner(s) without a formal medical examination by a healthcare provider. EPT has been legal in the state of Minnesota since 2008 (see Minnesota Statutes, Section 151.37 Subd. 2(g)) and may be prescribed by any licensed practitioner and/or another licensed healthcare professional under a collaborative practice agreement or protocol.
The potential public health benefits of EPT include:
- To reduce the number of reinfections and persistent infections
- To reduce complications associated with untreated STIs
- To decrease the probability of acquisition of other STIs including HIV
- To decrease overall antimicrobial exposure and thus, slow the development of antimicrobial resistance (AMR)
In an effort to address the increasing incidence of preventable sexually transmitted infections and their complications, the use of EPT in Minnesota is endorsed by the following agencies and professional organizations that are a part of the multidisciplinary medical community:
- Centers for Disease Control and Prevention
- American Medical Association
- American Osteopathic Association
- American Academy of Family Physicians
- American College of Obstetrics and Gynecology
- The Society for Adolescent Medicine
- American Academy of Pediatrics
- Minnesota Medical Association
- Minnesota Public Health Association
- Minnesota Society of Health-System Pharmacists
- Minnesota Pharmacists Association
- Minnesota Academy of PAs
- Minnesota Nurse Practitioners
- University of Minnesota College of Pharmacy
- University of Minnesota Medical School
While the ideal approach would include all partners being promptly notified of their exposure(s) and being evaluated, tested, and treated with preferred treatment regimens, this may not always be feasible. The CDC and/or MDH recommend that when partners of patients diagnosed with the following qualifying STIs are unable or unlikely to seek timely evaluation and treatment, EPT is recommended:
- Chlamydia
- Gonorrhea
- Trichomoniasis*
* The 2021 CDC STI guidelines note “EPT might have a role in partner management for trichomoniasis, however no partner management intervention has been demonstrated to be superior in reducing reinfection rates.” Some, but not all states offer EPT for infections due to T. vaginalis. Due to the potential consequences of untreated T. vaginalis infections, MDH allows licensed providers to prescribe EPT for trichomoniasis in Minnesota.
Published studies of EPT effectiveness have primarily included heterosexual individuals. There is less certainty of the effectiveness of EPT due to limited evidence and complexity in certain aspects of care in the following populations:
- Men who have sex with men (MSM)
- Adolescents*
- Pregnant women
*Minnesota Statutes, Section 144.343 says, "[a]ny minor may give effective consent for medical, mental and other health services to determine the presence of or to treat pregnancy and conditions associated therewith, venereal disease, alcohol and other drug abuse, and the consent of no other person is required), see the section entitled Providing EPT to Minors (under age 18) in Minnesota below.
EPT is permissible in the above populations, however healthcare clinicians should make a good faith effort to educate the index patient and their partner(s) about the importance of timely medical evaluation, testing, and treatment using preferred treatment regimens, and use their best judgment to determine whether EPT is appropriate.
MDH Antimicrobial Treatment Recommendations for EPT
Antimicrobial Treatment Regimens for Partners of Patients Diagnosed with Qualifying Sexually Transmitted Infections
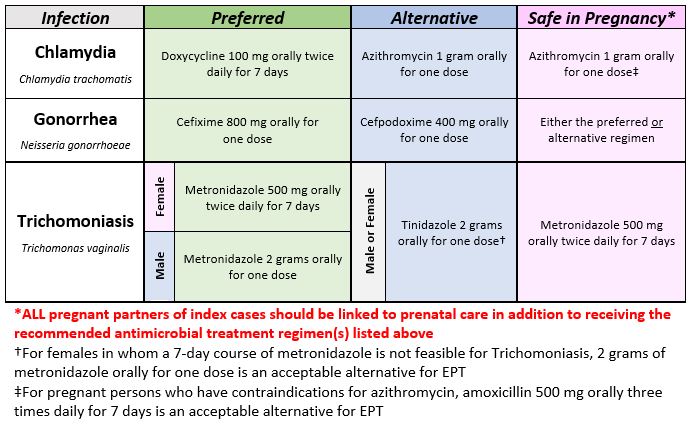
All patients should be educated to abstain from ANY sexual activity for 7 days after FINISHING their treatment regimen(s) even if their symptoms improve.
For a printable version of the above table, please refer to the EPT Regimen Quick Reference.
For recommendations on the management of index cases, please refer to the MDH STD section page and/or the 2021 CDC Sexually Transmitted Infection Treatment Guidelines.
Justification and Rationale
Epidemiology of Sexually Transmitted Infections Amenable to EPT in Minnesota
The epidemiology of STIs in Minnesota demonstrates the need for multiple strategies to reduce the incidence of sexually transmitted infections, including the use of EPT for partners who are unable or unlikely to seek timely treatment.
Gonorrhea
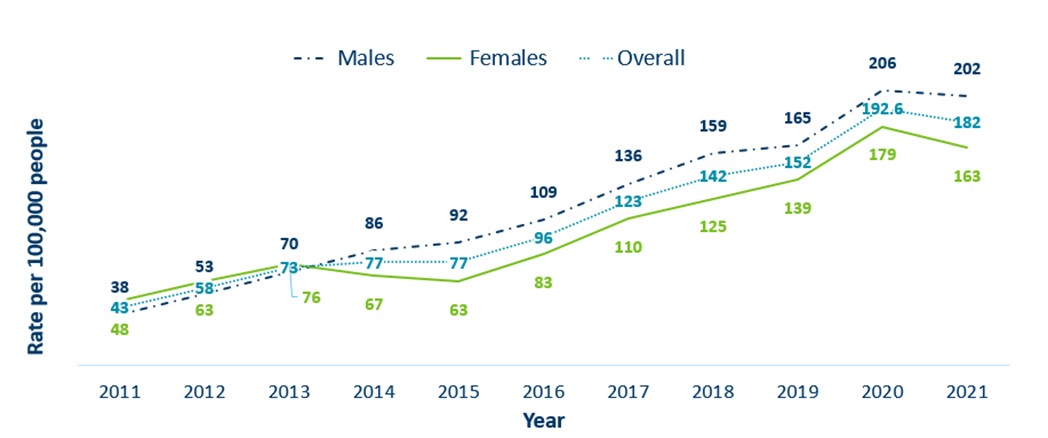
- The incidence of gonorrhea has increased by 423% in Minnesota from 2011 to 2021
- Since 2015, rates of gonorrhea amongst males and females have continued to increase proportionally year over year however males continue to have higher rates of gonorrhea in Minnesota
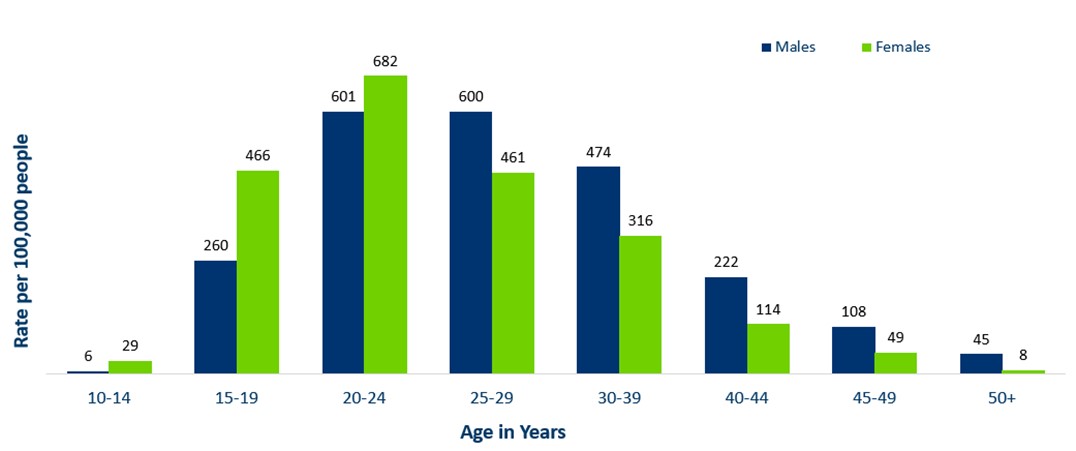
- Adolescents and young adults aged 15-24 make up only about 14% of Minnesota’s population, but account for 37% of all gonorrhea cases reported in 2021
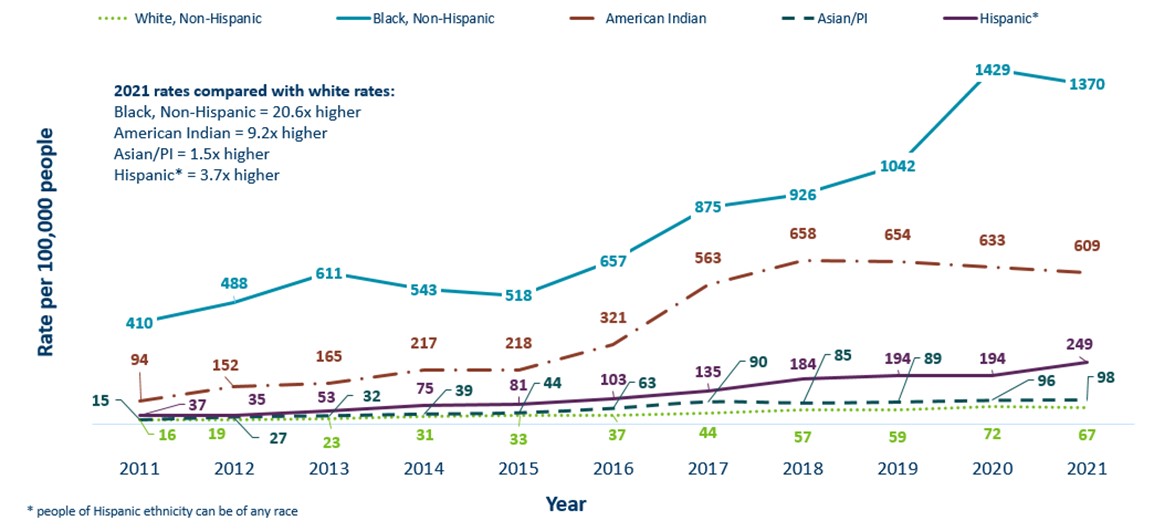
- Gonorrhea continues to disproportionately impact racial and ethnic groups most affected by the social determinants of health
Minnesota Incidence Rates of Gonorrhea by Gender Assigned at Birth, 2011-2021
Minnesota Incidence Rates of Gonorrhea by Age Group, 2021
Minnesota Incidence Rates of Gonorrhea by Race/Ethnicity, 2011-2021
Chlamydia
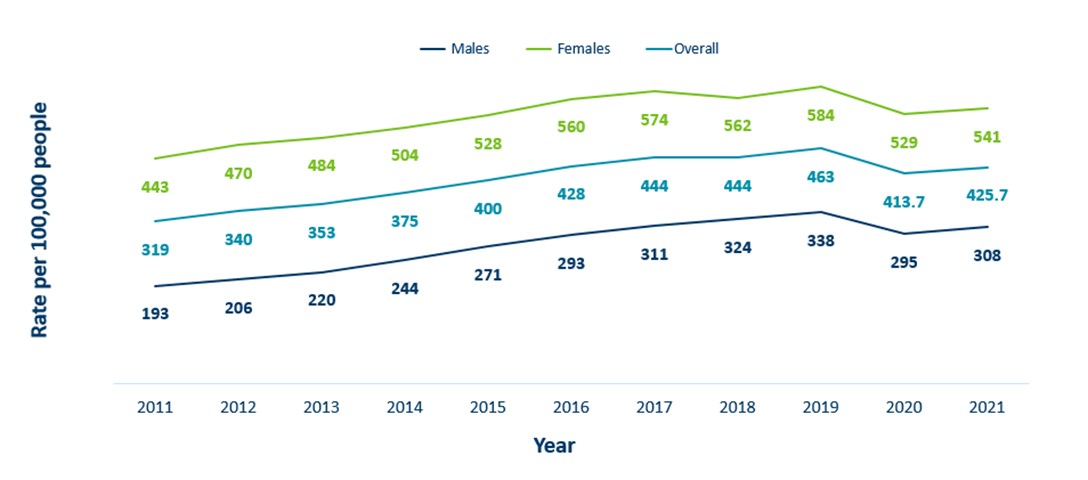
- The incidence of chlamydia has increased by 33% in Minnesota from 2011 to 2021
- Females continue to have significantly higher rates of chlamydia than males in Minnesota
- Despite the first observed decline in chlamydia incidence in over a decade recorded in 2020, incidence is again on the rise in 2021
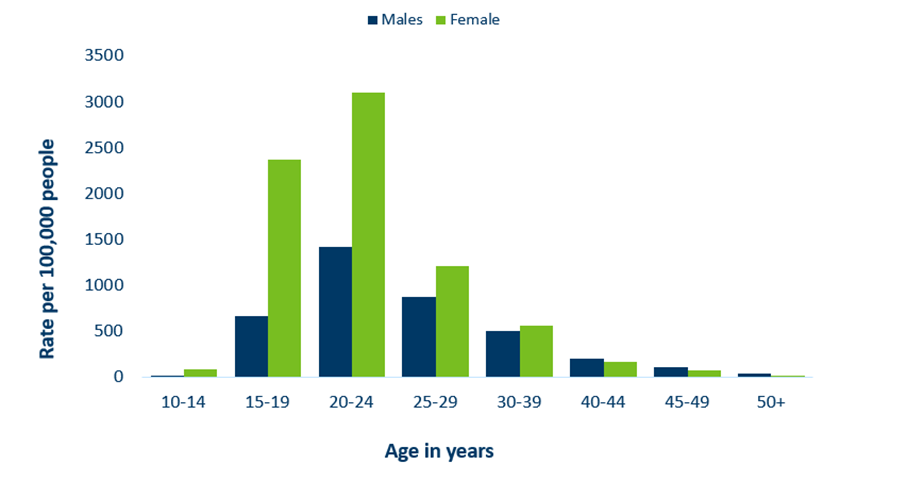
- Adolescents and young adults aged 15-24 make up only about 14% of Minnesota’s population, but account for 60% of all chlamydia cases reported in 2021
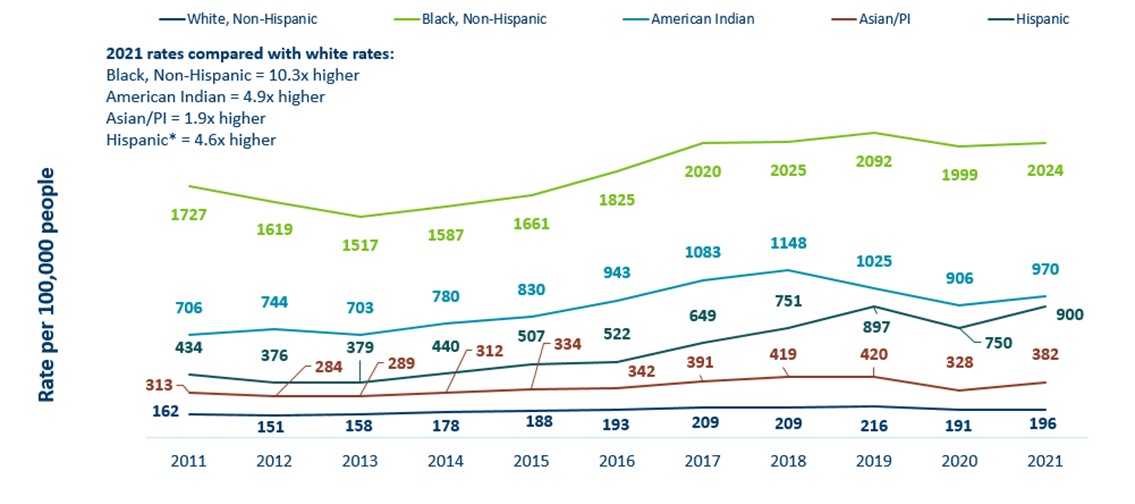
- Chlamydia continues to disproportionately impact racial and ethnic groups most affected by the social determinants of health
Minnesota Incidence Rates of Chlamydia by Gender Assigned at Birth, 2011-2021
Minnesota Incidence Rates of Chlamydia by Age Group, 2021
Minnesota Incidence Rates of Chlamydia by Race/Ethnicity, 2010-2020
Trichomoniasis
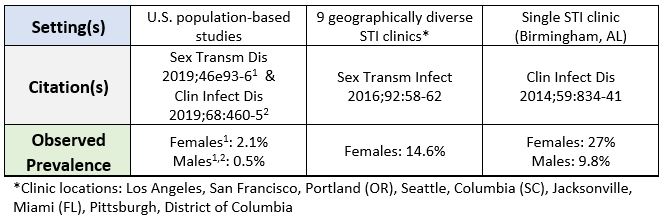
- As of 2022, Trichomoniasis is not a reportable disease in the United States or in Minnesota, so the local prevalence and incidence of T. vaginalis infections are not well understood. However, based on available population-based and surveillance studies:
- Estimated to be the most prevalent non-viral STI worldwide
- Estimated to affect approximately 3.7 million people in the United States
- Epidemiology likely varies widely based on several factors including: geographical region, gender assigned at birth, impact of the social determinants of health, among others
Population and Surveillance-based Estimates of Trichomoniasis Prevalence in the United States
Consequences of Untreated or Inadequately Treated Sexually Transmitted Infections
Personal
- Stigmatization
- Pelvic inflammatory disease (PID): approximately 40% of women with untreated chlamydia or gonorrhea may develop PID and increase risk for subsequent development of PID and complications
- Infertility, primarily in women but has rarely been reported in men as well: as many as 10% of women with their first episode of PID become infertile
- Ectopic pregnancy
- Increased risk of HIV acquisition via increased HIV shedding in persons living with HIV (PLWH) and/or increased host susceptibility
- Vertical transmission to neonates causing pneumonia, ocular, or other infections and complications
- Rarely, disseminated, or systemic infections in the case of certain STIs (e.g., gonorrhea, syphilis, HIV)
Population/Ecological
- Cycle of re-infection resulting in more complications, antibiotic use, and poorer quality of life
- Continue to widen health disparities and marginalization of those already most affected by the social determinants of health
- Increased economic burden and healthcare spending
- Promotion of antimicrobial resistance through increased antibiotic use
Staggering Economic Burden of Sexually Transmitted Infections in the United States
Based on an analysis of STI epidemiology in the United States in 2018 conducted by the CDC, lifetime medical costs from STIs (excluding costs associated with lost productivity, other non-medical costs, and STI prevention) are estimated as follows:
- $271 million attributed to new gonorrhea infections
- $691 million attributed to new chlamydia infections
- $144 million attributed to new Trichomonas infections
- Total of $1.106 billion in lifetime medical costs attributed these common, preventable infections
Estimated Cost of Sexually Transmitted Infections in the United States, 2018

Prescriber and Pharmacist Liability Relating to EPT
Since 2008 when EPT became legal in Minnesota, many stakeholders have worked together to ensure that EPT became the standard of care for partners of index patients diagnosed with qualifying sexually transmitted infections. As evidenced by the guidance herein, including the broad endorsement of the practice by numerous organizations representing the multidisciplinary medical community in Minnesota and throughout the United States, EPT is considered the standard of care in Minnesota. However, MDH cannot provide legal advice. Prescribers should check with their organization and/or own legal counsel on issues related to liability for potential injuries to partners resulting from EPT.
Please refer to the EPT Toolkit for Implementation in Clinical Settings for a more in-depth review of issues relating to prescriber liability.
Please refer to the EPT Toolkit for Minnesota Pharmacies for a more in-depth review of issues related to pharmacy and pharmacist liability.
Cost Considerations for EPT
Concerns surrounding the cost of EPT must be balanced by the adverse and costly health effects that may be averted by its use. EPT targets infected individuals who would otherwise not receive treatment. Therefore, widespread implementation of EPT would presumably reduce the number of infected persons in the population and lead to lower disease rates, thus reducing the number of patients needing costly treatment for long-term sequelae (e.g., PID, infertility, and ectopic pregnancy).
If personal information from the partner(s) is available:
- The EPT prescription may be run through that individual’s insurance as long as the prescription is in their name
- Antimicrobial treatment for STIs is generally well covered, however certain regimens (e.g., cefixime) may not be a part of the pharmacy benefit manager’s formulary
- If insurance information is available for partner(s), many EMRs can now report formulary status of medications being ordered prior to signing the prescription order
If personal information from the partner(s) is NOT AVAILABLE:
- The EPT prescription cannot be run through any type of insurance (e.g., using the index patient’s insurance with a refill for the partner is insurance fraud)
- Encourage the use of prescription coupon cards to reduce costs, if able
- Consider use of institutional or foundation grant funding for cases in which index patients and/or their partner(s) cannot afford to pay out of pocket for EPT
Approximate cost of common EPT regimens in Minnesota without insurance
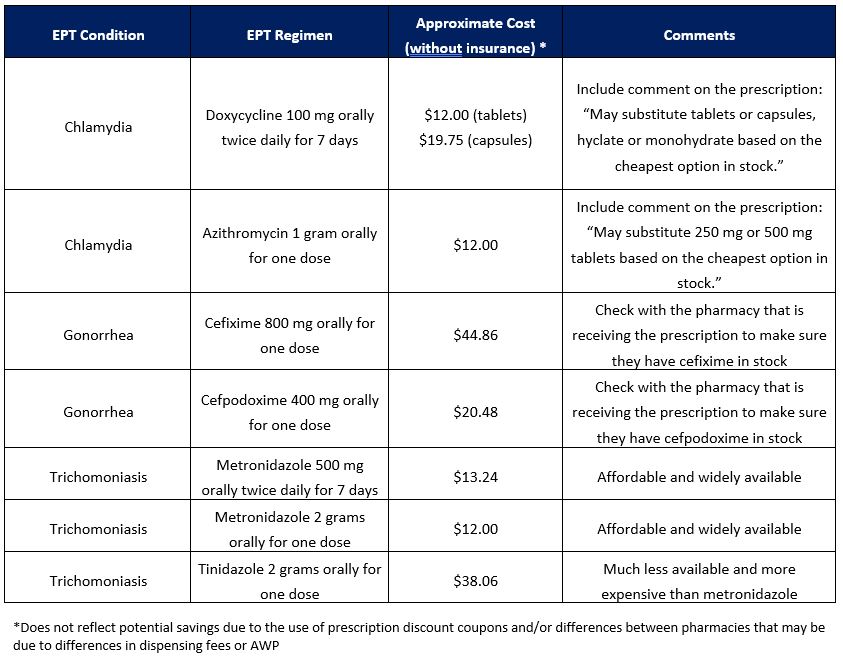
Pharmacy and Dispensing Requirements for EPT
EPT is considered standard of care and broadly endorsed by the interprofessional medical community in Minnesota, therefore prescriptions for EPT should be dispensed from Minnesota pharmacies when prescriptions are issued. Pharmacies are strongly encouraged to process, dispense, and education patients on EPT medications as outlined in MDH’s EPT guidance document (Minnesota Board of Pharmacy News, October 2018). Former Executive Director Cody Wiberg, PharmD, MS, RPh (2005-2022), stated “EPT prescriptions should be considered an order that may reasonably be dispensed by a Minnesota pharmacy, ideally with a name provided, but also without a partner name provided.”
- Under Minnesota Rule 6800.2250 Subp. 1(c): "[r]efusing to compound or dispense prescription drug orders that may reasonably be expected to be compounded or dispensed in pharmacies by pharmacists," (including EPT prescriptions) except as provided for in Minnesota Statutes, Sections 145.414 and 145.42 is unprofessional conduct and could result in disciplinary action by employer(s) and/or the Board of Pharmacy.
EPT Prescription Requirements (also see the EPT Prescription Requirement Quick Reference):

Prescribers are required to follow the requirements for a valid prescription as specified in Minnesota Statutes, Section 151.01 Subd. 16(a) with few allowable exceptions for EPT as shown in the example above and explained in bold below:
- The name of the location with address and phone number at which the EPT prescriber can be reached
- The date the EPT prescription is issued
- The name of the patient – for EPT: if the partner’s information is available, this should be included otherwise as shown above, it is NOT required in order to fill and dispense an EPT prescription according to former Board of Pharmacy executive director Dr. Cody Wiberg. Generic dummy names acceptable. The format of generic dummy names is flexible and will vary based on software capability. However, in order to make matching EPT prescriptions to the correct patient as easy as possible, best practice would be to include initials of the index patient (e.g., EPT Partner AB) in the dummy name. Prior to the EPT legislation passed in 2008, the patient’s name was required.
- The date of birth of the patient – for EPT: if the partner’s DOB is available, this should be included otherwise as shown above, it is NOT required in order to fill and dispense an EPT prescription. Blank, “n/a”, or generic dummy birthdays (e.g., 1/1/01) are acceptable.
- The address of the patient – for EPT: if the partner’s address is available, it should be included otherwise as shown above, it is NOT required in order to fill and dispense an EPT prescription. Blank, “n/a”, or generic dummy addresses (e.g., 111 EPT Drive, Minneapolis, MN 55404) are acceptable.
- For EPT: while not required, best practice is to indicate somewhere on the prescription that the intent is the issued prescription will be used for EPT. This indication would explain potentially missing information that would otherwise be required under the statute.
- The usual details about the drug being prescribed including full name of the drug including the drug strength, the “sig” or directions which should be as specific as possible to ensure the patient uses the medication properly, the quantity to dispense ideally written both numerically and alphabetically, and the number of refills – for EPT: refills are not allowed.
- Signature of the prescriber (either manual if it is a written prescription or electronic if it is an electronic prescription)
- The DEA is not required as none of the medications ordered for the purposes of EPT are controlled substances, however including the NPI number of the prescriber is recommended as a best practice (but not required)
For a printable version of the above figure and information, refer to the EPT Prescription Requirements Quick Reference.
According to the CDC, EPT prescriptions should also be accompanied with educational materials (refer to the Partner Education Materials section located at the bottom of this guidance) for the partnerthat discuss:
- Treatment instructions
- Warnings about medications
- General health counseling
- A statement advising that partner seek medical evaluation as soon as possible for HIV infection and any symptoms of STIs
For more in-depth pharmacy resources including sample pharmacy workflows, specific prescription questions, and pharmacy staff education, please refer to the EPT Minnesota Pharmacy Toolkit.
EPT Toolkits and Other Printable Materials
- EPT Toolkit for Minnesota Pharmacies
EPT Toolkit for Implementation in Clinical Settings
EPT Treatment Regimen Quick Reference
EPT Prescription Requirements Quick Reference
Printable Partner Education Materials
Your partner has Gonorrhea: What you need to knowYour partner has Chlamydia: What you need to know
Your partner has Trichomoniasis ("Trich"): What you need to know
Acknowledgments
This updated EPT guidance, including the comprehensive toolkits and quick reference documents, was made possible through the contributions of the following individuals and groups:
- Zachary Nelson, PharmD, BCPS, BCIDP, AAHIVP, MPH Candidate
- Candy Hadsall, MA, RN
- Patricia Segal Freeman, JD, MPH
- Khalid Bo-Subait, MPH
- Nick Lehnertz, MD, MPH, MHS
- Ruth Lynfield, MD
- Emily Groene Faherty, PhD
- Kelly Hadsall, PharmD
- Heidi Boulden, MSN, RN, PHN
- Emily Regan, MPH
- The MDH STD/HIV/TB Section
- The multidisciplinary medical community in Minnesota
