COVID-19
- COVID-19 Home
- Situation Update
- About COVID-19
- Testing
- Vaccine
- Protect Yourself & Others
- Medications
- Materials & Resources
- Stories of Community Outreach & Partnership
Information for
- Health Care
- Prisons, Detention Centers, & Homeless Service Sites
- Schools & Childcare
- Institutions of Higher Education
Related Topics
Contact Info
About COVID-19 Vaccine
 This webpage provides information to address the confusion surrounding the 2025-26 COVID-19 vaccine recommendations for Minnesotans. This includes where to get the COVID-19 vaccine and other vaccines to keep you and your family healthy.
This webpage provides information to address the confusion surrounding the 2025-26 COVID-19 vaccine recommendations for Minnesotans. This includes where to get the COVID-19 vaccine and other vaccines to keep you and your family healthy.
The U.S. Food and Drug Administration (FDA) approved four 2025–26 COVID-19 vaccines from three manufacturers on Aug. 27. For more detailed information on the vaccine products available visit Coronavirus (COVID-19): CBER-Regulated Biologics.
MDH endorses existing evidence-based guidance from independent experts and professional organizations such as the American Academy of Pediatrics (AAP), American Academy of Family Physicians (AAFP), American College of Obstetricians and Gynecologists (ACOG), and the Infectious Diseases Society of America (IDSA).
On this page:
2025–26 COVID-19 vaccine recommendations by group
What should you do
More information
2025–26 COVID-19 vaccine recommendations by group
| Group | Recommendation |
|---|---|
| Children 6 through 23 months | AAP: All children in this age group should be vaccinated with the 2025–26 COVID-19 formula, regardless of prior vaccination or infection history. Children should receive or complete the initial series. Children who are moderately or severely immunocompromised are recommended to have at least two doses depending on their vaccination history. Hospitalization rates in this group are high and are comparable to adults 50 to 64 years old. AAFP: Aligns with AAP. |
| Children 2 through 18 years | AAP: One dose recommended for certain children (those with underlying medical conditions, in long-term care/congregate settings, never vaccinated, or living with high-risk household members). For more information visit COVID-19 Vaccine Frequently Asked Questions. Vaccination may also be offered outside these groups if parents/guardians request. Children who are moderately or severely immunocompromised are recommended to have at least two doses depending on their vaccination history. AAFP: Aligns with AAP. |
| Adults 19 years and older | AAFP: All adults 19 years and older should be vaccinated, including pregnant women. This is especially important for adults 65 years of age and older, adults with medical conditions that increase risk of severe COVID-19, and those who were never vaccinated. Adults who are moderately or severely immunocompromised are recommended to have at least two doses depending on their vaccination history. |
| Adults 65 years and older | AAFP: Strong recommendation that all adults 65 years and older receive COVID-19 vaccination and a second dose 6 months later (minimum interval 2 months). Adults who are moderately or severely immunocompromised are recommended to have at least two doses depending on their vaccination history. |
| Pregnancy related | ACOG and AAFP: Recommend vaccination during pregnancy, when planning to become pregnant, in the postpartum period, or when lactating based on strong evidence of safety and protection for both the pregnant person and the baby. |
| People who are immunocompromised | IDSA: All immunocompromised children and adults should receive an age-appropriate dose of 2025-26 COVID-19 vaccine as soon as possible. Household members and people with close contact to the immunocompromised person should stay current with COVID-19 vaccination to reduce the risk of transmission. |
| Healthcare personnel | AAFP: Recommended vaccination who meet age requirement, lack documentation of vaccination or lack evidence of past infection. ACOG: Obstetrician–gynecologists and other healthcare practitioners are at high risk for exposure to COVID-19 and should lead by example by being vaccinated and encouraging eligible patients to be vaccinated as well. PALTmed: All clinical and non-clinical staff including vendors, contractors, and trainees in post-acute and long-term care (PALTC) facilities should receive one dose of the 2025-26 COVID-19 vaccine. AAFP: Adult Immunization Schedule (Table 2), ACOG: COVID-19 Vaccination Considerations for Obstetric–Gynecologic Care, and PALTmed: Recommendations for COVID-19 Vaccine in PALTC Residents and Staff. |
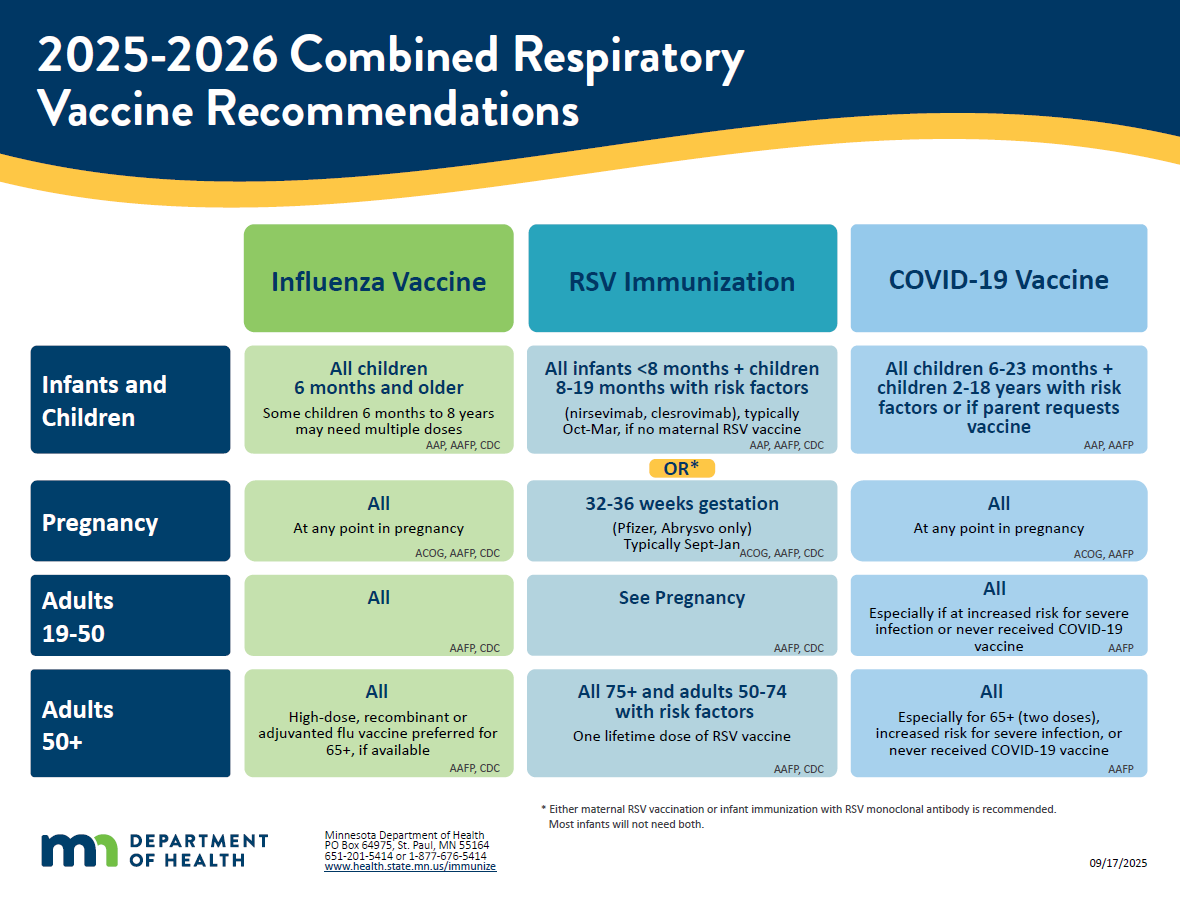 2025-2026 Combined Respiratory Vaccine Recommendations (PDF)
2025-2026 Combined Respiratory Vaccine Recommendations (PDF)
Handout version of the respiratory vaccine recommendations above.
What should you do
All Minnesotans over 6 months old are recommended to get COVID-19 vaccine if they want it. People 65 and older, those with underlying health conditions, young children, and pregnant people are strongly encouraged to get vaccinated. Minnesotans should talk with their healthcare provider to ensure they and their families are up to date. We recommend you contact your primary care provider or pharmacy in advance to ensure they have vaccine, and that you can receive one. If you are looking for vaccine for your child, start by checking with your child's primary care provider. Access for kids may take longer because there are fewer products this year and pharmacies cannot vaccinate children under the age of 3.
Find an immunization clinic near you. There are also resources for free or low-cost immunizations for people who do not have insurance or whose insurance does not cover the cost of vaccines.
More information
Fact sheets and videos
- Materials and Resources for COVID-19 Response: Vaccine
- Videos for COVID-19 Response: Vaccine
- Hais Txog COVID-19 (Hmong)
- Ku Saabsan KOFID-19 (Somali)
- Acerca del COVID-19 (Spanish)
Other websites
- COVID-19 Vaccine
Information, data, and guidance for COVID-19 vaccine in Minnesota. - Questions and Answers about COVID-19 Vaccines
Information from Children's Hospital of Philadelphia (CHOP) Vaccine Education Center.
